Beryllium (Be) has an atomic number of 4. This means it is the fourth lightest element, and 4th in the periodic table. More importantly, this means it has 4 protons in the nucleus. Find beryllium in the periodic table. Atomic number - 4. Atomic mass - 8. The atomic number = the number of protons Be has. Atomic mass = protons + neutrons.
- Beryllium Neutrons
- Beryllium Number Of Valence Electrons
- Beryllium Number Of Unpaired Electrons
- Number Of Protons Beryllium
Learning Objectives
- Define valence electron.
- Be able to indicate valence electrons when given the electron configuration for an atom.
Beryllium atoms have 4 electrons and the shell structure is 2.2. The ground state electron configuration of ground state gaseous neutral beryllium is He. 2s 2 and the term symbol is 1 S 0. The atomic number of each element increases by one, reading from left to right. Block Elements are organised into blocks by the orbital type in which the outer electrons are found. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f).
What makes a particular element very reactive and another element non-reactive?
A chemical reaction involves either electron removal, electron addition, or electron sharing. The path a specific element will take depends on where the electrons are in the atom and how many there are.
| Element Name | Symbol | Atomic Number | Electron Configuration |
| Lithium | Li | 3 | 1s22s1 |
| Beryllium | Be | 4 | 1s22s2 |
| Boron | B | 5 | 1s22s22p1 |
| Carbon | C | 6 | 1s22s22p2 |
| Nitrogen | N | 7 | 1s22s22p3 |
| Oxygen | O | 8 | 1s22s22p4 |
| Fluorine | F | 9 | 1s22s22p5 |
| Neon | Ne | 10 | 1s22s22p6 |
In the study of chemical reactivity, we will find that the electrons in the outermost principal energy level are very important and so they are given a special name. Valence electrons are the electrons in the highest occupied principal energy level of an atom. In the second period elements listed above, the two electrons in the 1 s sublevel are called inner-shell electrons and are not involved directly in the element's reactivity or in the formation of compounds. Lithium has a single electron in the second principal energy level and so we say that lithium has one valence electron. Beryllium has two valence electrons. How many valence electrons does boron have? You must recognize that the second principal energy level consists of both the 2 s and the 2 p sublevels and so the answer is three. In fact, the number of valence electrons goes up by one for each step across a period until the last element is reached. Neon, with its configuration ending in s2p6, has eight valence electrons.
Summary
- Valence electrons are the outer-shell electrons of an atom.
- Valence electrons determine the reactivity of an atom.
Practice
Use the link below to answer questions about valence electrons:
Review
- Define valence electron.
- Define inner shell electron.
- How many valence electrons are there in fluorine?
- What are the 2s electrons in nitrogen?
- How many inner shell electrons are there in beryllium?
Glossary
- inner-shell electrons: Those electrons that are not in the outer shell and are not involved in the reactivity of the element.
- valence electrons: The electrons in the highest occupied principal energy level of an atom.
References
- User:Chemicalinterest/Wikipedia. http://commons.wikimedia.org/wiki/File:Cobalt_carbonate.JPG.
Element Beryllium - Be
Comprehensive data on the chemical element Beryllium is provided on this page; including scores of properties, element names in many languages, most known nuclides of Beryllium. Common chemical compounds are also provided for many elements. In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies.
Beryllium Menu
- Beryllium Page One
- Beryllium Page Two
- Beryllium Page Three
Overview of Beryllium
- Atomic Number: 4
- Group: 2
- Period: 2
- Series: Alkali Earth Metals
Beryllium's Name in Other Languages
- Latin: Beryllium
- Czech: Beryllium
- Croatian: Berilij
- French: Glucinium
- German: Beryllium - s
- Italian: Berillio
- Norwegian: Beryllium
- Portuguese: Berílio
- Russian: Бериллий
- Spanish: Berílio
- Swedish: Beryllium
Atomic Structure of Beryllium
- Atomic Radius: 1.4Å
- Atomic Volume: 5cm3/mol
- Covalent Radius: 0.9Å
- Cross Section (Thermal Neutron Capture)σa/barns: 0.0092
- Crystal Structure: Hexagonal
- Electron Configuration:
- 1s2 2s2
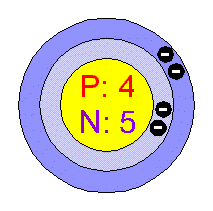
- Electrons per Energy Level: 2,2
- Shell Model
- Shell Model
- Ionic Radius: 0.35Å
- Filling Orbital: 2s2
- Number of Electrons (with no charge): 4
- Number of Neutrons (most common/stable nuclide): 5
- Number of Protons: 4
- Oxidation States: 2
- Valence Electrons: 2s2
- Electron Dot Model
- Electron Dot Model

Chemical Properties of Beryllium
- Electrochemical Equivalent: 0.16812g/amp-hr
- Electron Work Function: 4.98eV
- Electronegativity: 1.57 (Pauling); 1.47 (Allrod Rochow)
- Heat of Fusion: 12.2kJ/mol
- Incompatibilities:
- acids and strong bases, carbon tetrachloride, phos- phorous 3-chlorolithium, caustics, chlorinated hydrocarbons, oxidizers, molten lithium.
- Ionization Potential
- First: 9.322
- Second: 18.211
- Third: 153.893
- Valence Electron Potential (-eV): 82
Physical Properties of Beryllium
- Atomic Mass Average: 9.012182
- Boiling Point: 3243K 2970°C 5378°F
- Coefficient of lineal thermal expansion/K-1: 0.0000116E-6
- Conductivity
- Electrical: 0.313 106/cm Ω
Thermal: 2.01 W/cmK
- Electrical: 0.313 106/cm Ω
- Density: 1.848g/cc @ 300K
- Description:
- Strong, hard, gray-white metal. Lightest rigid metal. Formerly called glucinium (Gl).
- Elastic Modulus:
- Bulk: 110/GPa
- Rigidity: 156/GPa
- Youngs: 318/GPa
- Enthalpy of Atomization: 326.4 kJ/mole @ 25°C
- Enthalpy of Fusion: 11.72 kJ/mole
- Enthalpy of Vaporization: 294.7 kJ/mole
- Flammablity Class: Non-combustible solid (except as dust)
- Freezing Point:see melting point
- Hardness Scale
- Brinell: 600 MN m-2
- Mohs: 5.5
- Vickers: 1670 MN m-2
- Heat of Vaporization: 292.4kJ/mol
- Melting Point: 1551K 1278°C 2332°F
- Molar Volume: 4.88 cm3/mole
- Physical State (at 20°C & 1atm): Solid
- Specific Heat: 1.82J/gK
- Vapor Pressure 4.18kPa
Regulatory / Health
- CAS Number
- 7440-41-7 metal
- UN/NA ID and ERG Guide Number
- UN1567 / 134 powder
- RTECS: DS1750000
- NFPA 704
- Health: 4
- Fire: 1
- Reactivity: 1
- Special Hazard:
- OSHAPermissible Exposure Limit (PEL)
- TWA: 0.002 mg/m3
- Ceiling: 0.005 mg/m3
- Notes: Ceiling 0.025 mg/m3 30-min max peak
- OSHA PEL Vacated 1989
- TWA: 0.002 mg/m3
- Ceiling: 0.005 mg/m3
- Notes: Ceiling 0.025 mg/m3 30-min max peak
- NIOSHRecommended Exposure Limit (REL)
- Notes: Not to exceed 0.0005 mg/m3
- IDLH: 4 mg/m3 (Potential NIOSH carcinogen)
- Routes of Exposure: Inhalation; Skin and/or eye contact
- Target Organs: Eyes, skin, respiratory system
- Levels In Humans:
Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances.- Blood/mg dm-3: <1E-5
- Bone/p.p.m: 0.003
- Liver/p.p.m: 0.0016
- Muscle/p.p.m: 0.00075
- Daily Dietary Intake: 0.01 mg
- Total Mass In Avg. 70kg human: 0.036 mg
Beryllium Neutrons
Who / Where / When / How
- Discoverer: Nicholas Louis Vauquelin
- Discovery Location: Paris France
- Discovery Year: 1797
- Name Origin:
- From the mineral beryl.
- Abundance of Beryllium:
- Earth's Crust/p.p.m.: 2.6
- Seawater/p.p.m.:
- Atlantic Suface: 8.8E-08
- Atlantic Deep: 1.75E-07
- Pacific Surface: 3.5E-08
- Pacific Deep: 2.2E-07
- Atmosphere/p.p.m.: N/A
- Sun (Relative to H=1E12): 14
- Sources of Beryllium:
- Found mostly in minerals like beryl [AlBe3(Si6O18)] and chrysoberyl (Al2BeO4). Total world production per year is around 364 tons. Main mining areas are Brazil,USA, Madagascar, Germany, Czech Republic, Russia and India. Total world reserves estimated to be around 400,000 tons.
- Uses of Beryllium:
- Its ability to absorb large amounts of heat makes it useful in spacecraft, missiles, aircraft, etc. Emeralds are beryl crystals with chromium traces giving them their green color. Also used in light weight metal alloys, X-ray tube windows, watch springs and sparkless tools.
- Additional Notes:
- Beryllium was first discovered by Vauquelin in 1797, however, it was not issolated until 1828 in Berlin Germany and A.A.B bussy in Paris France. Beryllium comes from Greek beryllos, beryl. It has also been called Glucinium or Glucinum from the Greek word glykys which means 'sweet.' Beryllum is found in beryl, chrysoberyl and phenacite. Aquamarine and emeralds are precious forms of beryl (3BeO.Al2O3.6SiO2). It has a high melting point for a light metal and is more elastic than steel. It is used in computer parts, gyroscopes and for construction. Beryllium and its salts are toxic and should be handled with great care. Do not taste it to confirm that it is sweet. Beryllium is a potential occupational carcinogen (lung cancer).
Beryllium Menu
- Beryllium Page One
- Beryllium Page Two
- Beryllium Page Three
References
Beryllium Number Of Valence Electrons
A list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.
Related Resources
- Anatomy of the Atom
Answers many questions regarding the structure of atoms. - Molarity, Molality and Normality
Introduces stoichiometry and explains the differences between molarity, molality and normality. - Molar Mass Calculations and Javascript Calculator
Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. - Chemical Database
This database focuses on the most common chemical compounds used in the home and industry.
Citing this page
If you need to cite this page, you can copy this text:
Kenneth Barbalace. Periodic Table of Elements - Beryllium - Be. EnvironmentalChemistry.com. 1995 - 2021. Accessed on-line: 4/25/2021
https://EnvironmentalChemistry.com/yogi/periodic/Be.html/
.

.svg/953px-Electron_shell_004_Beryllium_(el).svg.png)
Linking to this page
If you would like to link to this page from your website, blog, etc., copy and paste this link code (in red) and modify it to suit your needs:
echo Periodic Table of Elements: Beryllium - Be (EnvironmentalChemistry.com)- Comprehensive information for the element Beryllium - Be is provided by this page including scores of properties, element names in many languages, most known nuclides and technical terms are linked to their definitions.
.
NOTICE: While linking to articles is encouraged, OUR ARTICLES MAY NOT BE COPIED TO OR REPUBLISHED ON ANOTHER WEBSITE UNDER ANY CIRCUMSTANCES.
Beryllium Number Of Unpaired Electrons
Chemical Properties of Beryllium
- Electrochemical Equivalent: 0.16812g/amp-hr
- Electron Work Function: 4.98eV
- Electronegativity: 1.57 (Pauling); 1.47 (Allrod Rochow)
- Heat of Fusion: 12.2kJ/mol
- Incompatibilities:
- acids and strong bases, carbon tetrachloride, phos- phorous 3-chlorolithium, caustics, chlorinated hydrocarbons, oxidizers, molten lithium.
- Ionization Potential
- First: 9.322
- Second: 18.211
- Third: 153.893
- Valence Electron Potential (-eV): 82
Physical Properties of Beryllium
- Atomic Mass Average: 9.012182
- Boiling Point: 3243K 2970°C 5378°F
- Coefficient of lineal thermal expansion/K-1: 0.0000116E-6
- Conductivity
- Electrical: 0.313 106/cm Ω
Thermal: 2.01 W/cmK
- Electrical: 0.313 106/cm Ω
- Density: 1.848g/cc @ 300K
- Description:
- Strong, hard, gray-white metal. Lightest rigid metal. Formerly called glucinium (Gl).
- Elastic Modulus:
- Bulk: 110/GPa
- Rigidity: 156/GPa
- Youngs: 318/GPa
- Enthalpy of Atomization: 326.4 kJ/mole @ 25°C
- Enthalpy of Fusion: 11.72 kJ/mole
- Enthalpy of Vaporization: 294.7 kJ/mole
- Flammablity Class: Non-combustible solid (except as dust)
- Freezing Point:see melting point
- Hardness Scale
- Brinell: 600 MN m-2
- Mohs: 5.5
- Vickers: 1670 MN m-2
- Heat of Vaporization: 292.4kJ/mol
- Melting Point: 1551K 1278°C 2332°F
- Molar Volume: 4.88 cm3/mole
- Physical State (at 20°C & 1atm): Solid
- Specific Heat: 1.82J/gK
- Vapor Pressure 4.18kPa
Regulatory / Health
- CAS Number
- 7440-41-7 metal
- UN/NA ID and ERG Guide Number
- UN1567 / 134 powder
- RTECS: DS1750000
- NFPA 704
- Health: 4
- Fire: 1
- Reactivity: 1
- Special Hazard:
- OSHAPermissible Exposure Limit (PEL)
- TWA: 0.002 mg/m3
- Ceiling: 0.005 mg/m3
- Notes: Ceiling 0.025 mg/m3 30-min max peak
- OSHA PEL Vacated 1989
- TWA: 0.002 mg/m3
- Ceiling: 0.005 mg/m3
- Notes: Ceiling 0.025 mg/m3 30-min max peak
- NIOSHRecommended Exposure Limit (REL)
- Notes: Not to exceed 0.0005 mg/m3
- IDLH: 4 mg/m3 (Potential NIOSH carcinogen)
- Routes of Exposure: Inhalation; Skin and/or eye contact
- Target Organs: Eyes, skin, respiratory system
- Levels In Humans:
Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances.- Blood/mg dm-3: <1E-5
- Bone/p.p.m: 0.003
- Liver/p.p.m: 0.0016
- Muscle/p.p.m: 0.00075
- Daily Dietary Intake: 0.01 mg
- Total Mass In Avg. 70kg human: 0.036 mg
Beryllium Neutrons
Who / Where / When / How
- Discoverer: Nicholas Louis Vauquelin
- Discovery Location: Paris France
- Discovery Year: 1797
- Name Origin:
- From the mineral beryl.
- Abundance of Beryllium:
- Earth's Crust/p.p.m.: 2.6
- Seawater/p.p.m.:
- Atlantic Suface: 8.8E-08
- Atlantic Deep: 1.75E-07
- Pacific Surface: 3.5E-08
- Pacific Deep: 2.2E-07
- Atmosphere/p.p.m.: N/A
- Sun (Relative to H=1E12): 14
- Sources of Beryllium:
- Found mostly in minerals like beryl [AlBe3(Si6O18)] and chrysoberyl (Al2BeO4). Total world production per year is around 364 tons. Main mining areas are Brazil,USA, Madagascar, Germany, Czech Republic, Russia and India. Total world reserves estimated to be around 400,000 tons.
- Uses of Beryllium:
- Its ability to absorb large amounts of heat makes it useful in spacecraft, missiles, aircraft, etc. Emeralds are beryl crystals with chromium traces giving them their green color. Also used in light weight metal alloys, X-ray tube windows, watch springs and sparkless tools.
- Additional Notes:
- Beryllium was first discovered by Vauquelin in 1797, however, it was not issolated until 1828 in Berlin Germany and A.A.B bussy in Paris France. Beryllium comes from Greek beryllos, beryl. It has also been called Glucinium or Glucinum from the Greek word glykys which means 'sweet.' Beryllum is found in beryl, chrysoberyl and phenacite. Aquamarine and emeralds are precious forms of beryl (3BeO.Al2O3.6SiO2). It has a high melting point for a light metal and is more elastic than steel. It is used in computer parts, gyroscopes and for construction. Beryllium and its salts are toxic and should be handled with great care. Do not taste it to confirm that it is sweet. Beryllium is a potential occupational carcinogen (lung cancer).
Beryllium Menu
- Beryllium Page One
- Beryllium Page Two
- Beryllium Page Three
References
Beryllium Number Of Valence Electrons
A list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.
Related Resources
- Anatomy of the Atom
Answers many questions regarding the structure of atoms. - Molarity, Molality and Normality
Introduces stoichiometry and explains the differences between molarity, molality and normality. - Molar Mass Calculations and Javascript Calculator
Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. - Chemical Database
This database focuses on the most common chemical compounds used in the home and industry.
Citing this page
If you need to cite this page, you can copy this text:
Kenneth Barbalace. Periodic Table of Elements - Beryllium - Be. EnvironmentalChemistry.com. 1995 - 2021. Accessed on-line: 4/25/2021
https://EnvironmentalChemistry.com/yogi/periodic/Be.html/
.
Linking to this page
If you would like to link to this page from your website, blog, etc., copy and paste this link code (in red) and modify it to suit your needs:
echo Periodic Table of Elements: Beryllium - Be (EnvironmentalChemistry.com)- Comprehensive information for the element Beryllium - Be is provided by this page including scores of properties, element names in many languages, most known nuclides and technical terms are linked to their definitions.
.
NOTICE: While linking to articles is encouraged, OUR ARTICLES MAY NOT BE COPIED TO OR REPUBLISHED ON ANOTHER WEBSITE UNDER ANY CIRCUMSTANCES.
Beryllium Number Of Unpaired Electrons
Number Of Protons Beryllium
PLEASE, if you like an article we published simply link to it on our website do not republish it.
